
Analytical Writing for Creative Thinking
It’s Easier Than You Think, Really.
Curious Learner
For as long as I can remember, from my earliest memories, I have always been a curious individual who loves to learn about everything around me. Much to the annoyance of my teachers, but thanks to my parents who encouraged that behavior, I developed a habit of diving deep into various topics, especially science, a practice I still maintain today. Overtime I wanted the desire to express creative ideas, with accurate knowledge gave rise to analytical writer.
None of your organs work in isolation; they are interconnected with other systems that together create the overall function of the body. This concept appears everywhere: from the well being of individual communities, to the delivery of healthcare, to the performance of national economies, and even to how we think about the purpose of the universe.
This can also be said for medical writing. An exceptional medical writer can not only learn new topics at an almost inhuman pace, but they can also critique and credit well written scientific papers while communicating to a wide range of audiences without spreading misinformation. This is something a hallucinating AI cannot replace, especially when many documents are used for payers or regulatory purposes that impact access to healthcare.


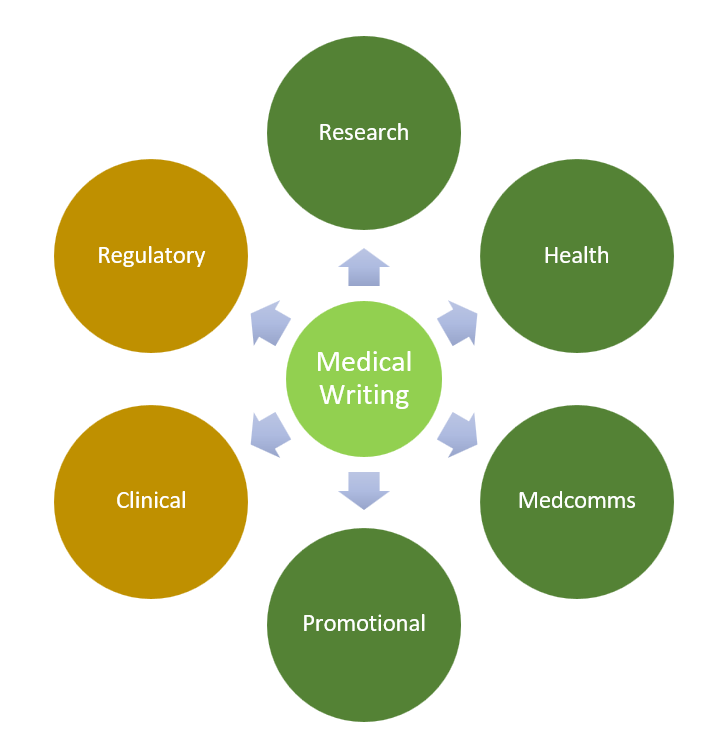
Research
A way for science to express itself. Also called scientific papers or academic papers. It contributes to new information or verifies what we already know. The two most common are research papers (you execute and collect original data) and review papers (you evaluate other people’s data). A medical writer helps draft the paper to ensure the writing is clear, concise, and accurately interpreted. They also ensure that all citations and references are trackable and formatted according to the publisher’s requirements.
Health
This type of expression is used for educational purposes, specifically for the general public and patients who do not have a robust understanding of medical information. Medical writers simplify complex information to educate about diseases, treatment options, and wellness guidance that accurately presents the evidence. They play a vital role in educating patients about any risks and side effects they may experience so they can make informed decisions.
Medcomms
This is also for educational purposes, tailored for healthcare professionals (HCPs), regulators, researchers, medical affairs teams, and clinicians. Medical writers are able to assess study design, examine the sample size and endpoint selection, critique statistical methods, and identify any biases to validate data and demonstrate the effectiveness of new treatments or therapies.
Promotional
This form of writing incorporates visual elements to encourage the use of a new treatment or therapeutic option. It is not the typical sales pitch most people imagine, as it still needs to be scientifically accurate. Medical writers must understand the impact of disease beyond signs and symptoms in order to write patient brochures that reduce reluctance. They also need to summarize a 50‑page clinical document into a single poster to validate data for prescribing decisions for HCPs, and create effective slide decks that compare new treatments with existing options for payers.
Clinical
This form of technical writing requires strict adherence to deadlines and ICH guidelines for FDA/EMA. Clinical Study Reports (CSRs) describe methods and interpret results of clinical trials. The Statistical Analysis Plan (SAP) is a blueprint for how clinical trial data will be analyzed. Investigator Brochures (IBs) are updated reference documents with all essential information such as safety, dosing, and risks about the drug. Informed Consent Forms (ICFs) facilitate the process of obtaining informed consent from patients who are willing to participate. Case Report Forms (CRFs) collect patient data, including age, medical history, outcomes, and adverse events. As a medical writer, you will be contributing to most of these documents filed together as clinical trial documents.
Regulatory
This form of technical writing focuses on getting new drugs and therapies approved in accordance with FDA/EMA requirements. Investigational New Drug (IND) applications contain preclinical data, the clinical protocol, and information on the drug’s mechanism, and are submitted to the FDA before a clinical trial begins. New Drug Applications (NDAs) or Marketing Authorization Applications (MAAs) are final submissions requesting approval for the drug to enter the market. Risk Management Plans (RMPs) and Risk evaluation and Mitigation (REMS) outline any known side effects, potential risks, and missing information to support risk‑minimization strategies.
Medical Writing
A medical writer is a blend of a technical writer, someone who drafts formal, specialized documents and user manuals, and a creative writer, who crafts more informal content using different voices. Both skill sets are applied to topics related to medicine. Some writers become highly specialized in a specific niche, commonly in regulatory or clinical areas, while others, like myself, prefer to be well rounded to satisfy a natural curiosity. Regardless of the path you choose, it is essential to remain accurate to the science.
Unfortunately, the U.S. Bureau of Labor Statistics does not provide verifiable data specific to medical writing. As a result, most information about career trajectories in this field is anecdotal. However, according to Grand View Research, the global pharmaceutical market is projected to reach $2.776 trillion by 2033. As the industry expands, the demand for regulatory and clinical writers is expected to grow. In addition, there will be an increasing need for creative medical communicators who can educate clinicians, patients, and payers in clear and engaging ways.
One may still be skeptical with A.I taking over the role. A.I is a large language model (LLM) trained with large set of data found online and copy right material (this is why we see many lawsuit against major A.I company). The reason it can not replace medical writers is due to the following.
Misinformation: This happens for two major reasons. First, most of the data used to train large language models comes from the internet, and fact‑checkers already struggle with the accuracy of online information. Second, AI can hallucinate, meaning it generates the next sequence of words based on patterns and user prompts rather than verified facts. In general, AI performs well with broad, common knowledge but often gets complex or specialized medical information wrong.
Poor communicator: When used as an editor, it adds unnecessary words or stretches simple information, making it confusing, especially for the general public or patients who don’t have the same level of understanding as a researcher or clinician. Simple and straight to the point is the best approach.
Skimming: When using it as a search engine to find relevant papers, it regularly misses results, especially if they are behind a paywall. It also misses a lot that can still be found by digging more into PubMed. This can misrepresent data and delay regulatory documents.
Terrible for our environment: AI data centers require large amounts of electricity to keep the software running and need clean water to cool it down. This has unfortunately raised utility bills for locals who cannot keep up with the rising cost of living. There are a limited number of RAM units manufactured, with data centers outcompeting gaming console and PC developers. This is also raising the cost of day‑to‑day electronics.
Types of Medical Writing
Services Tailored for You
Due to my extensive experience and deep understanding of scientific literature developed through academic achievement and personal research projects, I am able to provide high‑quality writing services that fully align with your objectives. For writing samples, please visit the blog section or my LinkedIn profile. All posts are fully cited, thoroughly researched, and not AI‑generated, offering genuine insight and original analysis. I also write nonfiction pieces at Creativebrosis.
I am available for one‑time or multi‑project work as an independent contractor or ghostwriter in the following areas:
Drafting, summarizing, and fact‑checking research papers with proper references for clinical trials or primary research studies.
Creating easy‑to‑understand brochures, educational blogs, and social media content tailored for general audience, clinician, stakeholder and collaborators.
Writing peer‑reviewed manuscripts with strict adherence to publisher guidelines.
Developing educational materials as books, posters, templates and newsletters.
Designing visually engaging slide decks with personalized animations created in Krita and DaVinci.
I am currently independently studying regulatory and clinical documentation and will begin offering regulatory‑focused services once I enroll in and complete the UC San Diego Medical Writing Certificate program in Fall 2026. I am also open to volunteer or internship opportunities in this field and fully enroll in a PhD program by 2027 to advance my career as a well‑rounded medical writer


For general inquiries, don’t forget to…
info@analyticalwriter.com


Bitter Reality


The way we use automation is very interesting. When it is applied to repetitive tasks, it works wonders and saves a tremendous amount of time. But when automation is used as a creative outlet through blogs, videos, or infographics, it creates an echo chamber of misinformation and recycled content. This gives us the false illusion that we live in an age of abundant factual information. With AI, the automation of creative content is happening at lightning speed, causing everything to sound the same. At the same time, valuable information that requires time, expertise, and intensive focus becomes buried by algorithms that prioritize high volume output rather than high quality publication.
It is easy to blame AI, the people who created it, the people using it, or even to label me as anti‑AI, which I am not. My focus is simply to bring awareness. AI can be an exceptional tool for specific tasks, such as predicting protein folding or finding relevant research papers through tools like Research Rabbit, because these systems are designed around factual data rather than pattern recognition.
We need to ensure that the use of AI is ethical by making data publicly accessible and by getting consent from the creators whose work is used in training datasets. This may not be possible for large tech companies, but we can support organizations that choose to operate transparently. As an artist who has spent twenty years developing my craft, it is disheartening to see artists’ work taken and used to generate poorly drawn infographic templates that contribute to the same echo‑chamber content. Instead, we can use free online tools like SmartArt in Microsoft Word or even make a quick sketch and upload it ourselves.
Relying on AI‑generated content does not only harm creative expression; it also harms you, because you miss the opportunity to learn the skills required to create meaningful and valuable work.
If you appreciate our work, even when you are not using our services, that support means a lot. If this message resonates with you, please share our articles and connect with me through LinkedIn. Thank you for your time and understanding.

